Quick Facts
- Category: Health & Medicine
- Published: 2026-05-04 12:15:47
- Solateria Breaks the Mold: Difficulty Options Don't Sacrifice Challenge in Soulslike Metroidvania
- Exploring Python 3.13's Enhanced Interactive REPL: A Comprehensive Guide
- Transform Your Google Home Mini into a Home Assistant Hub: A Step-by-Step Guide
- FDA Moves to Restrict Compounded Versions of Popular Weight Loss and Diabetes Drugs
- J. Craig Venter: The Scientist Who Revolutionized Genomics and Defied Expectations
High cholesterol remains a leading cause of heart disease, but a revolutionary new approach is offering hope beyond traditional statins. Researchers have engineered tiny DNA molecules that target and disable PCSK9, a protein that prevents the liver from removing harmful LDL cholesterol. This therapy has shown remarkable results, reducing bad cholesterol by nearly 50% in early trials. Whether you're statin-intolerant or simply seeking an alternative, understanding this innovation is crucial. Below, we explore the seven key things you need to know about this game-changing treatment.
1. What Exactly Is PCSK9 and Why Does It Matter?
PCSK9 (proprotein convertase subtilisin/kexin type 9) is a protein produced primarily in the liver that regulates LDL cholesterol levels. Normally, LDL receptors on liver cells bind to and remove bad cholesterol from the blood. However, PCSK9 attaches to these receptors and marks them for destruction, reducing the liver's ability to clear LDL. This leads to higher cholesterol levels and increased risk of arterial plaque buildup. The new DNA-based treatment works by silencing the PCSK9 gene, effectively stopping the production of this troublesome protein. By doing so, it allows LDL receptors to remain active and pull more cholesterol out of circulation, mimicking a natural genetic variation that some people have and that protects against heart disease.
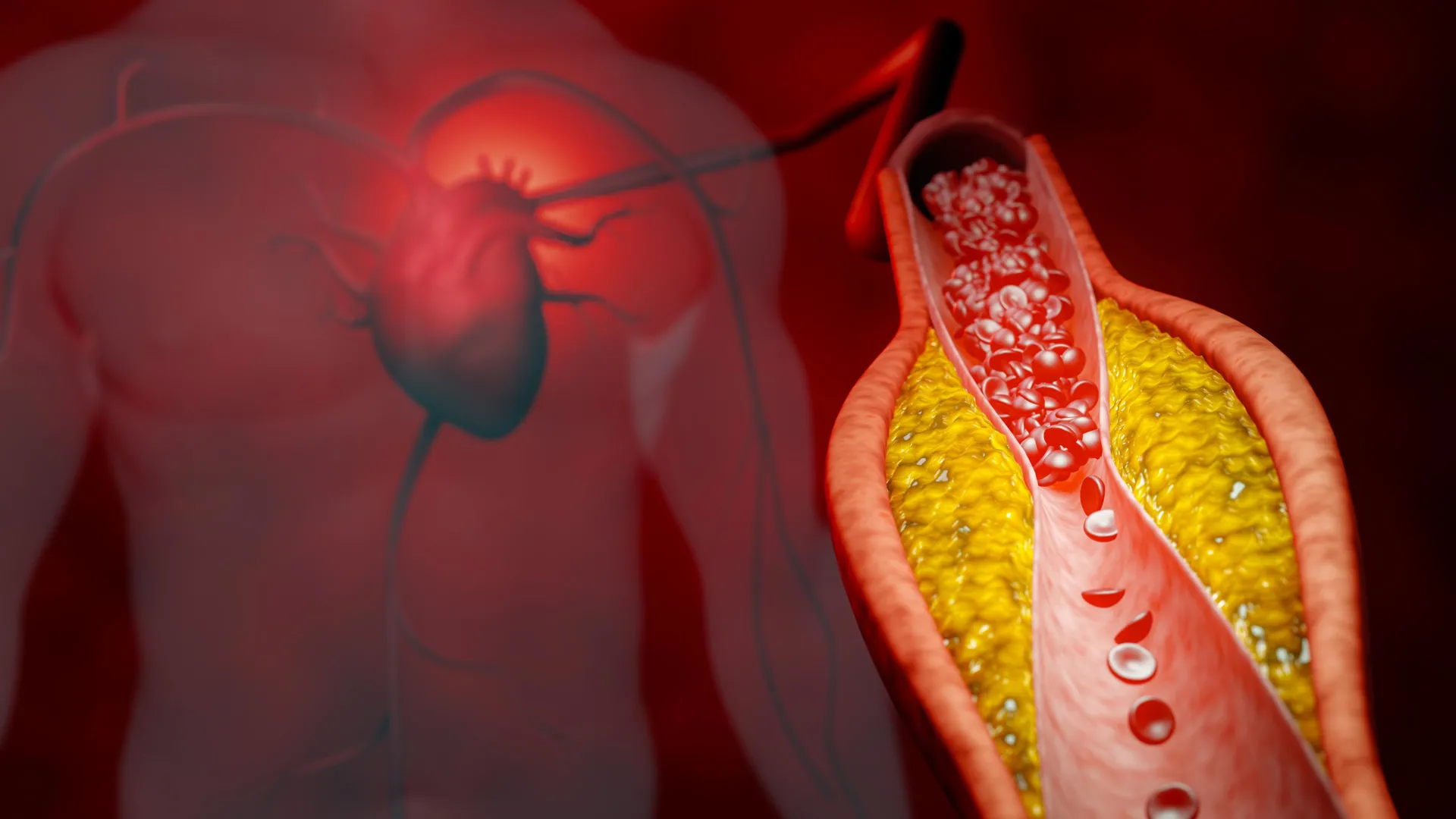
2. How Do Tiny DNA Molecules Shut Down a Protein?
The treatment uses antisense oligonucleotides (ASOs)—short, synthetic strands of DNA designed to bind to the messenger RNA (mRNA) that carries the instructions for making PCSK9. Once bound, the ASO triggers the cell's own machinery to degrade that mRNA, preventing the protein from ever being assembled. This is a form of gene silencing that is highly specific. Unlike traditional drugs that block a protein after it's made, ASOs nip the problem in the bud. The therapy is administered via injection, typically every few weeks, and the effects last long enough to maintain steady LDL reduction. Because the molecules are DNA-based, they are biodegradable and have a low risk of accumulation in tissues, making them a promising alternative to monoclonal antibodies that also target PCSK9 but are more expensive and require more frequent dosing.
3. Promising Clinical Results: Nearly 50% Reduction in LDL
In a phase 2 clinical trial, patients receiving the ASO therapy experienced an average 49% reduction in LDL cholesterol compared to placebo, with some individuals seeing drops of over 60%. These results were achieved on top of standard statin therapy, meaning the DNA molecules provided an additive effect. Importantly, the reduction occurred without the safety concerns often associated with high-dose statins, such as muscle pain or liver enzyme elevations. The trial enrolled people with high cardiovascular risk, including those with familial hypercholesterolemia. The durability of the effect is notable—the LDL levels remained suppressed even after discontinuation of the treatment, suggesting that a short course could have lasting benefits. These findings have sparked excitement among cardiologists, who see it as a potential game-changer for patients who cannot attain target LDL levels with existing medications.
4. A Statin-Free Alternative for Intolerant Patients
Statins are the gold standard for lowering LDL, but up to 10–20% of patients experience side effects like myalgia (muscle pain), fatigue, or increased blood sugar. For these individuals, the new DNA-based therapy offers a lifeline. Because it works through a completely different mechanism—suppressing PCSK9 synthesis rather than inhibiting an enzyme in the liver—it bypasses the pathways that cause statin intolerance. Moreover, it does not interfere with the production of CoQ10, a compound often depleted by statins and linked to muscle symptoms. Early data suggest that the ASO is well tolerated, with only minor injection-site reactions and no serious adverse events in the trials. This could expand treatment options for millions of patients who currently avoid or stop statins, leaving them vulnerable to heart attacks and strokes.
5. How It Compares to Existing PCSK9 Inhibitors
Currently, two monoclonal antibodies—alirocumab (Praluent) and evolocumab (Repatha)—are FDA-approved as PCSK9 inhibitors. These are injected every two to four weeks and can lower LDL by 50–60%. However, they are extremely expensive (often over $14,000 per year) and require cold chain storage. The new ASO therapy offers several advantages: it is cheaper to manufacture, stable at room temperature, and may require less frequent dosing as studies explore a once-monthly or even once-quarterly schedule. Additionally, ASOs are easier to modify for sustained release. The DNA molecules also have a lower immunogenicity risk compared to antibodies, meaning the body is less likely to develop resistance over time. While both approaches effectively lower cholesterol, the ASO platform could make advanced cholesterol management more accessible and affordable, potentially reducing healthcare disparities.
6. Who Is the Ideal Candidate for This New Treatment?
The therapy is being developed primarily for high-risk patients who cannot achieve LDL goals with maximally tolerated statins, including those with atherosclerotic cardiovascular disease, familial hypercholesterolemia, or diabetes. It may also be appropriate for people with a history of statin intolerance or those who have contraindications to statins. However, it is not intended for everyone with high cholesterol. Lifestyle modifications like diet and exercise remain first-line therapy. Patients with very high baseline LDL (above 190 mg/dL) or those with genetic forms of high cholesterol, such as heterozygous familial hypercholesterolemia, could benefit significantly. As trials progress, the therapy may also be studied for primary prevention in younger adults with moderately elevated LDL, but that is likely years away. For now, the focus is on high-risk populations where the absolute benefit is greatest.
7. What Does the Future Hold for DNA-Based Cholesterol Therapies?
The success of this PCSK9-targeting ASO paves the way for a new class of genetic therapies for cardiovascular disease. Researchers are already exploring similar approaches for other lipid-related targets, such as Lp(a) and angiopoietin-like proteins. Moreover, the ASO platform could be adapted for other metabolic conditions. The next steps include larger phase 3 trials to confirm efficacy and safety, followed by regulatory submissions. If approved, the therapy could be available within 3–5 years. Challenges remain, including ensuring equitable access and managing the logistics of injectable therapies. However, the promise is clear: a durable, well-tolerated, and potentially cost-effective way to lower cholesterol without statins. This breakthrough not only changes how we treat high cholesterol but also opens the door to personalized, gene-based approaches that could revolutionize cardiovascular care.
Conclusion: A New Era in Cholesterol Management
The development of DNA-based molecules that shut down PCSK9 represents a paradigm shift in cardiovascular medicine. By offering a nearly 50% reduction in bad cholesterol without the side effects of statins, this therapy addresses a critical unmet need for millions of patients. As with any innovation, cost and accessibility will determine its real-world impact, but the scientific foundation is solid. Whether you are a patient seeking alternatives or a clinician expanding your toolkit, staying informed about these advances is essential. The days of one-size-fits-all cholesterol treatment are numbered—welcome to the era of precision cardiology.